You can be certain that SAMSON AKTIENGESELLSCHAFT is well informed about the Regulation (EC) No. 1907/2006 (REACH), which entered into force on 1 June 2007, and has determined the applicable consequences and obligations.
On 1 September 2008, the European Chemicals Agency (ECHA) first published a REACH candidate list according to Article 59 (1, 10) and Article 33. It includes chemical substances and serves as the basis for the authorization or restriction of substances depending on their use.
Substances are constantly being added to the SVHC list (substances of very high concern). As a result, it is an on-going process to check whether our products contain substances of very high concern (SVHC) in a concentration greater than 0.1 % (w/w).
We apply the O5A (once an article, always an article) approach to every (formerly) individual article specified on a bill of materials. We are in close contact with our suppliers as part of this process.
We manufacture "articles" as defined in the REACH Regulation; as a result, we are a "downstream user". Consequently, we have a duty to communicate information to our customers on substances contained in our products according to the REACH Regulation.
Since January 2021, we submit this information to the European Chemicals Agency (ECHA). It publishes this information to support waste operators in improving waste management and recycling practices.
ECHA makes this information available on the SCIP database (Substances of Concern In Products), https://echa.europa.eu/de/scip-database.
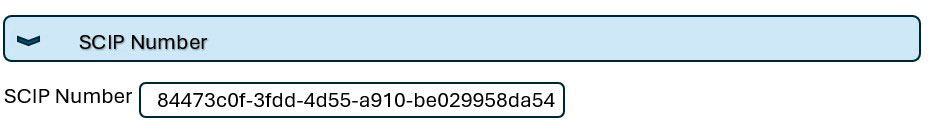
Each dataset for a product that SAMSON AKTIENGESELLSCHAFT submits has its own SCIP number.
You can find affected SAMSON products in the SCIP database either by clicking on the Search button (highlighted below) and entering the SAMSON product details as the article identifier:



Click on icon to obtain more information on the substance contained in the product and instructions on its safe use.
Document "Additional Information on Your Inquiry/Order"
You are informed of the SCIP number in the quotation or order documents (Article 33(1) in REACH). We do not provide confirmation in any other data formats since the SCIP data are sufficient to meet the legal requirements.
As a result of legal amendments to REACH Annex XVII (Annex 17), some products are subject to a restriction, which only allows commercial use under controlled conditions. These amendments are taken into account in the information that we submit to ECHA and that we communicate to our customers. You will be informed if SAMSON products contain substances that are subject to the REACH authorization process according to Annex XIV (Annex 14). Based on the information we have from our supply chains, we currently do not have any evidence of this.
Downloads
Helpful links
- REACH: what is it?
National REACH helpdesks inside and outside the EU Information portal of the European Chemicals Agency ECHA - Help Desk, Health and Safety Executive in the United Kingdom